|
While these names are distinctive and, therefore, are considered chemically-correct, the common system has two significant limitations. Various bonding configurations of transition-metal atoms on carbon nanotubes: Their effect on contact resistance Antonis N. For example, the Latin stem for "iron" is "ferr-." Therefore, using the common system, Fe 2 is called the "ferrous ion," and Fe 3 is named as the "ferric ion." The bond made by electron sharing is called a covalent bond. These shared electrons simultaneously occupy the outermost shell of both atoms. The second way for an atom to obtain an octet of electrons is by sharing electrons with another atom. In most cases, the stem, or main portion, of the elemental name used in the common system is typically derived from the element's Latin name. The resulting compounds are called ionic compounds. The "-ous" suffix indicates the cation with the lesser relative cation charge, and an "-ic" suffix is used to refer to a cation with a larger charge. Metal carbonyls are compounds where the metal and carbon bond (M-C) has both s and p character. The second system, called the common system uses two suffixes, "-ous" and "-ic," to distinguish the names of transition metal elements. 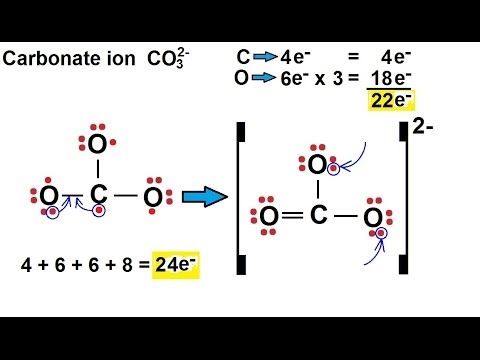 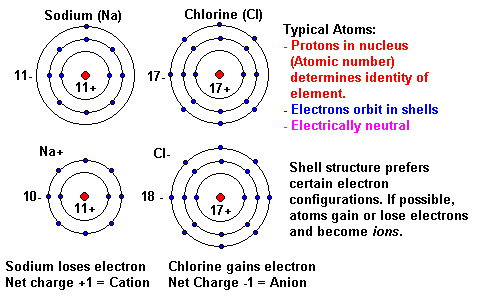 
\): Possible Transition Metal Charges and Their IUPAC Roman Numeral Designations Charge
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
